Mental Health Innovation: Why Psychedelic Therapies are Attracting Big Pharma?
Psychedelic therapies are witnessing an exponential surge in the mental health space. This resurgence stems from simplified or relaxed regulatory barriers, shifts in public perception, and the lack of new psychopharmacological options in the psychiatry pipeline. Additionally, rigorous scientific research on psychedelic drugs and evidence of efficacy from clinical trials are also adding to their mainstream acceptance.
The psychedelic drug market is forecasted to grow by $8.8B USD in 2031, with a CAGR of 13.3% since 2025. This market valuation emphasizes the lucrative nature and popularity of psychedelics in the pharma sector. Because they can produce effective, rapid, and stable changes within a few sessions, psychedelic-assisted therapies are now highly recognized, especially as mental health disorders like depression, anxiety, and PTSD put immense pressure on healthcare.
However, intellectual property issues, the need for long supervised sessions, and the efficiency of clinical testing results in comparison to lab trials create roadblocks for medical industry players in the widespread adoption of psychedelic therapies in mental health treatment or mood disorders.
Emerging Psychedelic Innovations Redefining the Pharmaceutical Landscape
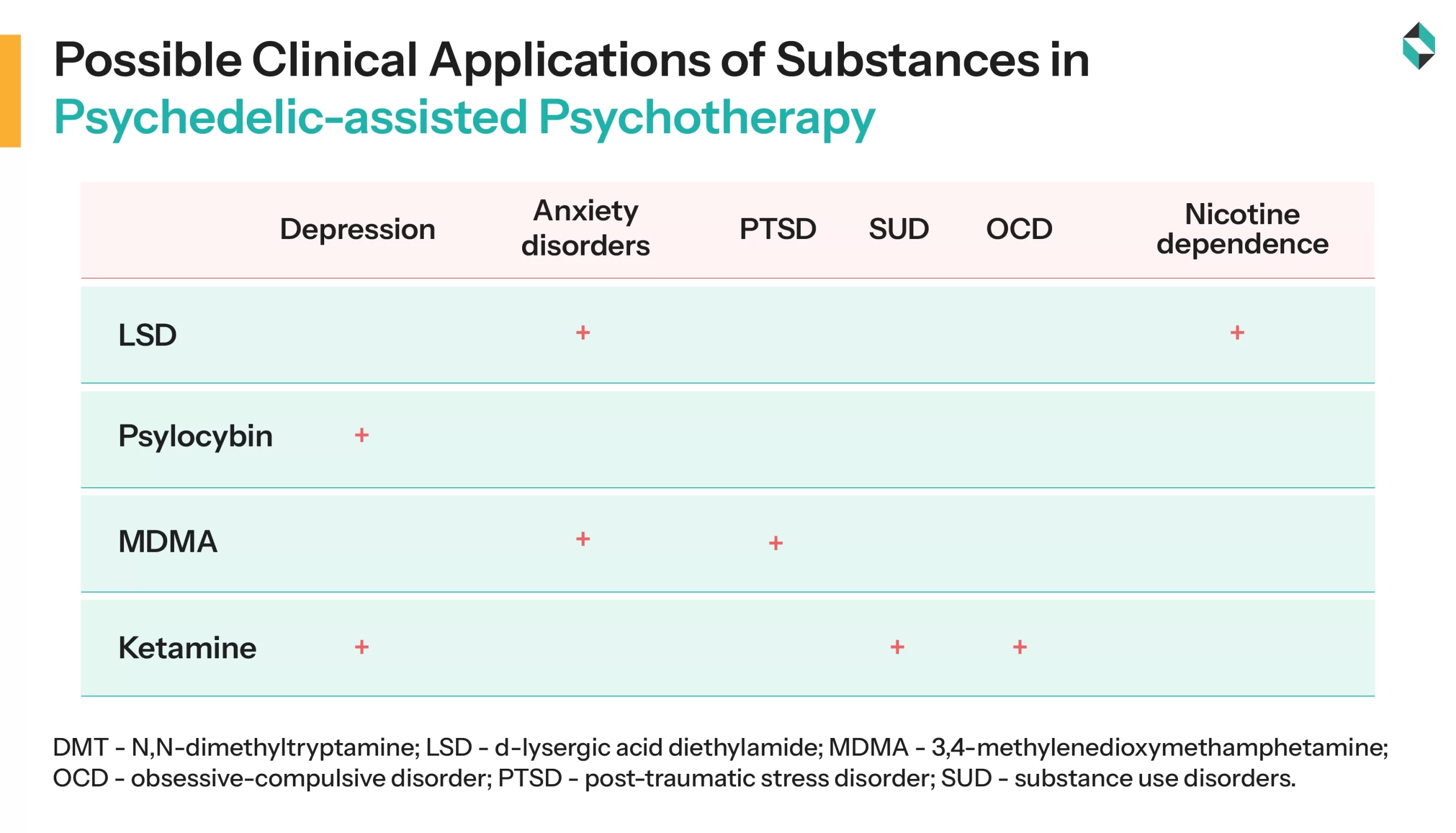
Pharma innovators are extensively involved in studying compounds like DMT, MDMA, LSD, and psilocybin to examine their potential in treating mental health issues. The researchers and regulators are also directing efforts towards making psychedelics commercially viable and scientifically validated.
Several industry leaders, including Compass Pathways, Cybin, and Atai Life Sciences, are developing synthetic forms of psilocybin and R-Ketamine, respectively. COMP360 is a synthetic form of psilocybin developed and patented by Compass Pathways. It is the active compound in magic mushrooms. This innovation is currently in phase 2 trials for PTSD and phase 3 trials for depression, mainly treatment-resistant depression.
Subsequently, Cybin is utilizing deuterated molecules to develop a synthetic psilocybin analogue, CYB003. The primary goal of this innovation is to ensure a brief duration and rapid onset of psychedelic therapy, making it viable for healthcare settings by reducing the amount of supervision needed for patients.
Furthermore, MM-120, BPL-003, RE104, and EMP-01 are emerging next-generation psychedelics designed to provide lasting relief and reduce the cost of treating mental health disorders. They are considered to be effective in treating conditions ranging from generalized anxiety and major depression to alcohol use disorders.
Why Psychedelic Therapy is Gaining Momentum in the Mental Health Space among Pharmaceuticals
New business models for revenue generation and increased effectiveness are key factors drawing pharmaceutical companies toward psychedelic therapy. There is a move toward highly effective sessions combined with long-term remission, with companies focusing on short, manageable trip durations to fit standard outpatient clinic workflows.
Pharmaceutical businesses and stakeholders can capitalize on market opportunities, including unmet needs in treating anxiety, PTSD, and TRD, by harnessing the rapid and sustained benefits of psychedelics. For example, Otsuka Pharmaceutical’s acquisition of Mindset Pharmaceutical aims to fill a market gap and reach millions of treatment-resistant patients.
High-value psychedelics are changing the mental health innovation landscape as the focus shifts to business models based on interventional sessions rather than daily pill taking. Additionally, as companies experiment with 5-MeO-DMT and other short-acting compounds to reduce session durations from 1-2 hours to 6-8 hours, the main goal is to promote precision in treatment and scalability of business frameworks for clinical workflows.
Improved neuroplasticity and long-term effectiveness are key benefits of psychedelic therapy, as data highlights that a short treatment or a single dose can manage mental health disorders for months, compared to standard Selective Serotonin Reuptake Inhibitors (SSRIs) that often require months or weeks.
Overall, the strong cognitive benefits, rapid outcomes, and favorable safety profile of psychedelic-assisted therapies are attracting market players and encouraging exploration of this market’s potential.
Critical Bottlenecks for Pharmaceutical Companies in Using Psychedelic Therapies for Mental Health Treatment
1. Scalability and Operational Barriers: The need for long professional supervision, high labor and cost intensity due to 1:1 or 2:1 supervised administration, and substantial infrastructural requirements pose scaling bottlenecks.
2. Regulatory Complexities: Most Serotonergic Psychedelic compounds remain Schedule I and need adequate clinical evidence and strategic legal management for moving toward FDA approval.
Blinding difficulties and the high standards set by the FDA, despite strong data, indicate that the pharma industry needs to bridge the gap between real-world feasibility and clinical trials. The case of Lykos Therapeutics reflects this, as the FDA rejected a request for approval of MDMA combined with therapy for treating patients struggling with treatment-resistant PTSD.
3. Ethical Risks: Commercial Determinants of Health (CDoH), where the profit intent may conflict with public health objectives, increasing the chances of downplaying risks of psychedelic medicines and the dissemination of misleading advertisements or sponsored messages.
4. Clinical & Safety Risks: There is a lack of understanding regarding the long-term implications of experimental approaches, which necessitates the set inclusion/exclusion criteria that may pose safety risks. Additionally, there is uncertainty about managing potential destabilizing experiences or psychosis-like symptoms that occur outside of the clinical setting.
What are the Strategic Implications for Pharma Stakeholders?
- Business stakeholders need to fulfill the prerequisites of having a PAT-trained workforce and relevant infrastructure for conducting psychedelic therapy sessions for commercial feasibility and scalability.
- With strong growth projections of the psychedelic market and rising demand for alternative treatments over conventional ones, there is a need to navigate the conflicting interests between providing access and benefiting the public through profit-generating models. Moreover, a balance between commercialization and medicalization is largely required.
- In order to shift to routine care and the commercialization of psychedelic support for mental health treatment, legislative support and adequate reimbursement rates are needed to incentivize healthcare providers and insurance firms.
- A proper care ecosystem where policymakers, psychologists, and scientists work closely together is the way forward for the successful integration of psychedelic medicines in treating mental health disorders.
- Standardization and service-based revenue streams can be profitable for big pharma, where business models should incorporate specialized clinical infrastructure alongside medications.
- Organizations indulging in M&A tend to secure IP in patented molecules, thereby future-proofing their growth. For example, the extensive partnerships and acquisition efforts of Johnson & Johnson and Otsuka clearly demonstrate this trend.
Final Words
The strategic landscape is shifting towards interventional and integrated medicines, with a greater focus on long-term patient outcomes rather than the traditional daily-pill approach. Although there are significant regulatory hurdles and the need for specialized infrastructure and therapist training, successful phase 3 trials and the rising number of FDA breakthrough designations are helping to legitimize these psychoactive substances.
Success for market players and big pharma stakeholders depends on aligning their research with independent validation and credible evidence to ensure long-term authenticity within clinical communities. The coming years will determine whether the healthcare and pharma industry can overcome ethical and other significant obstacles to advance psychedelic-assisted therapies into safe, effective, inclusive, and evidence-based care from specialized trials.
Our pharmaceutical consulting experts at Stellarix assist companies in overcoming R&D challenges, regulatory complexities, and risks in their business and innovation roadmaps. From strengthening the innovation portfolio and providing insights on cutting-edge technologies to evidence-generation tactics, our skilled team offers actionable guidance to pharma companies to stay ahead in a changing industrial landscape through our R&D services.
Let's Take the Conversation Forward
Reach out to Stellarix experts for tailored solutions to streamline your operations and achieve
measurable business excellence.