FRESH Act 2026: Decoding the Tale of Two Food Industries
Key Takeaways:
- As the state-by-state patchwork wipes out, compliance costs come down, but also erase the clean-label, ban-based market differentiation.
- The bill inclines towards the rediscovery of pre-1958 ingredients.
- While large manufacturers gain speed and scale from this act, small and mid-level players lose a key market-shaping tool.
The Dual-Impact Proposition
The FDA’s Review and Evaluation for Safe, Healthy, and Affordable Foods Act, or the FRESH Act bill, arrives at a precarious moment for food manufacturers. The scenario resulting from this is both rewarding and complicated. On the one hand, its unified national regulatory framework simplifies operations and growth for large multi-state food manufacturers; it takes away the power of market differentiation and flexibility of small and mid-level manufacturers that they enjoyed through a state-by-state approach.
The FRESH Food Act Bill: The Core Pillars of Its Impact
The impact of this development is rooted in four fundamental provisions. Before we move to understand its strategic impact, it is essential to take a brief look at them.
- The Revised Definition of Ingredients: The act signals a two-tier, non-flexible framework of food ingredients.
- The first tier exempts ingredients normally sold or consumed as food on or before Jan 1, 1958, from being considered as food additives. It includes a wide range of substances, going from fruits & vegetables to meat, algae, bacterial cultures, and their derivatives.
- The second tier mandates a complete shift from the present voluntary GRAS notification system and makes it mandatory for all, along with a public registry.
- The Preemption Sledgehammer: One of the most debatable but consequential levers for food giants, it effectively wipes out all state laws related to the sale, labeling, use, and marketing of dietary and food supplements. It essentially targets the growing legal state-level bans on specific chemicals like PFAS in packaging and artificial colors in school meals.
- The Novel, Industry-Embedded Review Pathway: The bill formally acknowledges third-party scientific panels for the evaluation of GRAS determinations. It is estimated that it might be used as a “rubber stamp” by industry-sponsored groups to weaken FDA oversight.
- Infant Formula and Post-Market Safety Provision: From September 2026, the bill mandates a step-by-step post-market food chemical safety review program, along with a long public list of chemicals prioritized for risk examination next year. It gives the FDA the final recall authority for adulterated infant formula and allows it to restrict toxic elements in baby food.
Decoding its Strategic Impact
The FRESH Act reshuffles the complete regulatory framework set for food manufacturers with some clear pros and cons.
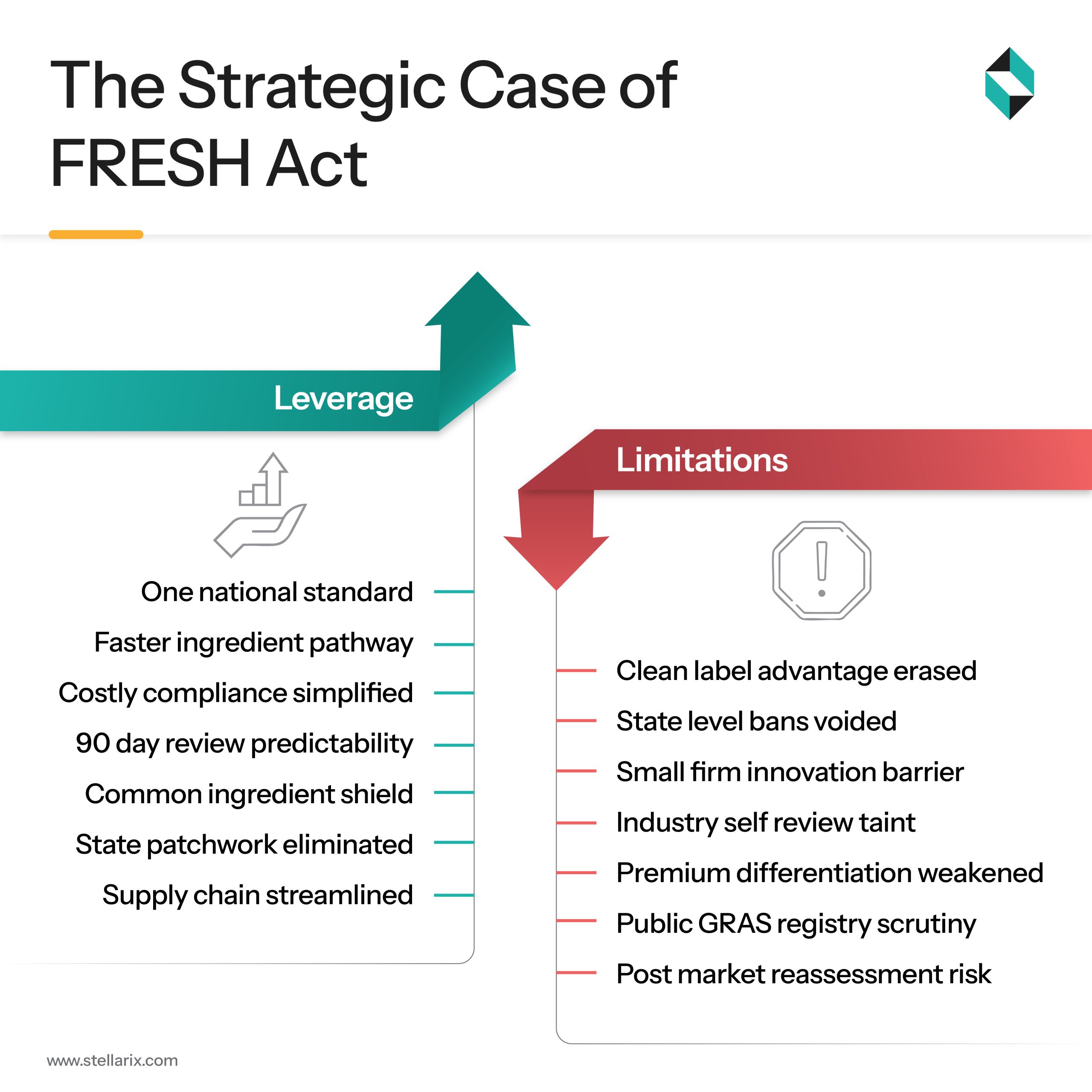
The Pros: National Uniformity and Operational Efficiency
This bill is clearly a big win for multi-state food manufacturers. It provides them with one thing they were striving for long, predictability, and operational simplicity. They can now overcome the supply chain inefficiencies and regulatory complications resulting from multiple state-level bans on titanium dioxide, PFAS, etc. The “Common Food Ingredients” category resulting from this bill promises regulatory certainty to a huge portfolio of conventional, commodity-based entities.
The Cons: Reducing Consumer Trust and Market Power
Au contraire, smaller brands that were building their business by offering “cleaner than the standard” labels see this bill as strategic headwinds. The preemption clause prevents them from entering premium, revenue-generating, trend-aligned categories. The state-wise regulatory floor that padded their offering by positioning them as “free from” certain banned ingredients is now gone. Also, the industry-funded review will erode consumer trust by fading transparency in truly novel and independently scrutinized ingredients.
The Innovation Impact
The new bill not just shifts the focal point of R&D initiatives, but also redirects their flow. The architecture of this act is more inclined towards specific innovations while reducing the appeal of a few others.
- Revitalizing Traditional Platforms with “Common Food Ingredient”: The permanent exemption of ingredients used before 1958 is a huge innovation incentive. It will compel manufacturers to revise their R&D strategies. They can now reallocate their resources to rediscovering and commercializing existing solutions rather than discovering completely new molecules.
- Faster Cycles, Fewer Moon Shots: The accelerated but transparent GRAS pathway is a two-edged sword for innovators. While a clocked review creates a time-bound, predictable process that reduces months from GTM processes. The transparency filter will increase scrutiny from competitors, specialized media, and NGOs. The funnel for innovation will be narrow at the top and will allow only well-documented, robust scientific dossiers.
- Post-market Reassessment: This program directly challenges the ongoing “introduce and defend approach to novel ingredients. It will facilitate the “safety-by-design” innovation philosophy with better upfront investments in bio-accumulation and long-term toxicology studies. In the long term, it will also raise the R&D cost of entry for de novo ingredients, resulting in a barrier favoring science-driven, well-capitalized companies.
Bottomline
This act incentivizes the shift of food innovation from a high-risk, high-reward gamble to a disciplined process of perfecting, verifying, and scaling the ingredients that are already there. It is a huge win for large-sized, science-backed companies that have resources to navigate the new regulatory systems, but it complicates the path of smaller, trend-oriented organizations that will have to find new ways to position and differentiate themselves in this market.
Wondering if the FRESH act will be a tailwind or a roadblock for your portfolio? Our consulting team at Stellarix is helping companies turn regulatory disruptions into a strategic advantage by building strategic blueprints that pave the path to the right side of the fork. Connect with us to map your innovation pipeline.
Let's Take the Conversation Forward
Reach out to Stellarix experts for tailored solutions to streamline your operations and achieve
measurable business excellence.



