Redefining Psychiatry: Psychedelic Neurotherapeutics as a Clinical Paradigm Shift
What’s Driving the Renewed Interest in Psychedelic Medicine?
The increasing acceptance of psychedelic neuro-therapeutics is changing mental health care while eliminating harm for the most difficult-to-treat malaise of our times: anxiety, depression, addiction, PTSD, etc. Encouraging early results with psychedelics like psilocybin, MDMA, and others have spurred the opening of clinics offering them, and will continue to gain traction and reshape mental health treatment in the future. Here’s what’s driving the shift.
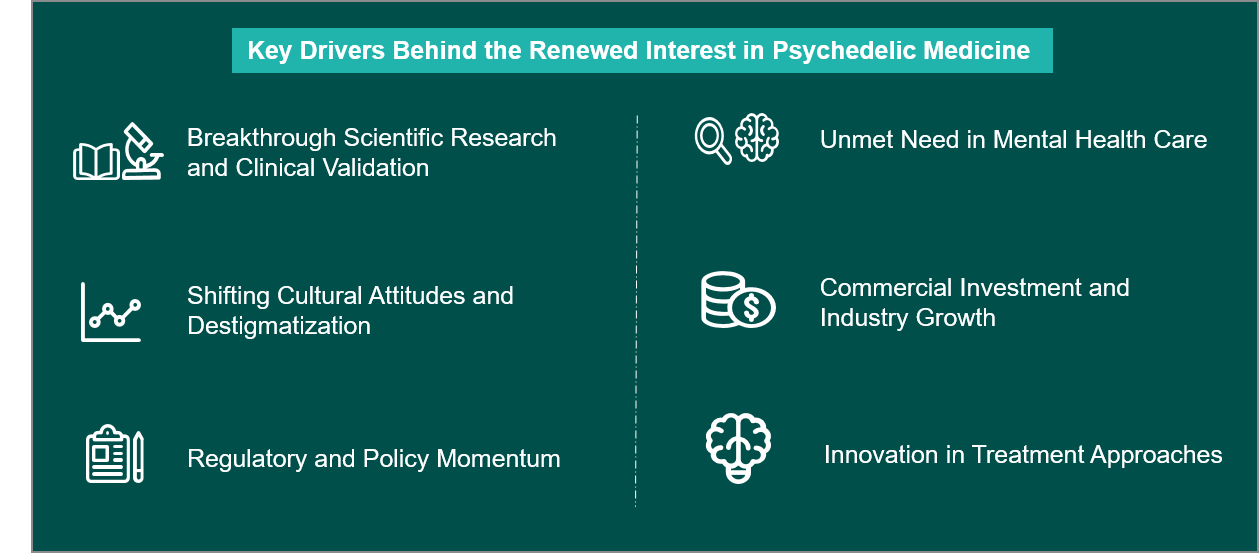
The Regulators Are Paying Attention – And That Changes Everything: Regulators are now providing more support for psychedelic research by providing structure and fast-tracking development. Because of this, there is less uncertainty in the development process, which means more businesses participating in the psychedelic research sector.
Science Is Rewriting the Narrative: As stigma continues to erode, evidence from research studies and increased media coverage has contributed to the normalization of psychedelics within culture. As a result, trends show improved patient recruitment for studies, increased willingness from practitioners to use these types of treatments, and an overall more favorable environment for changes in laws and the commercialization of these products.
It’s Not Just a Drug – It’s an Experience: Psychedelics are introducing a hybrid model that combines drug therapy with guided psychotherapy, and this integrated approach focuses on long-term behavioral and emotional change rather than symptom management alone.
Rising Consumer Awareness
Increased interest in wellness trends such as microdosing has brought psychedelics into mainstream conversation. While still evolving, this trend is expanding awareness and future market potential.
From Classic Psychedelics to Cutting-Edge Breakthroughs
Psychedelics target specific centers of the brain to alter their understanding of sensory input. They work on the brain in different ways:
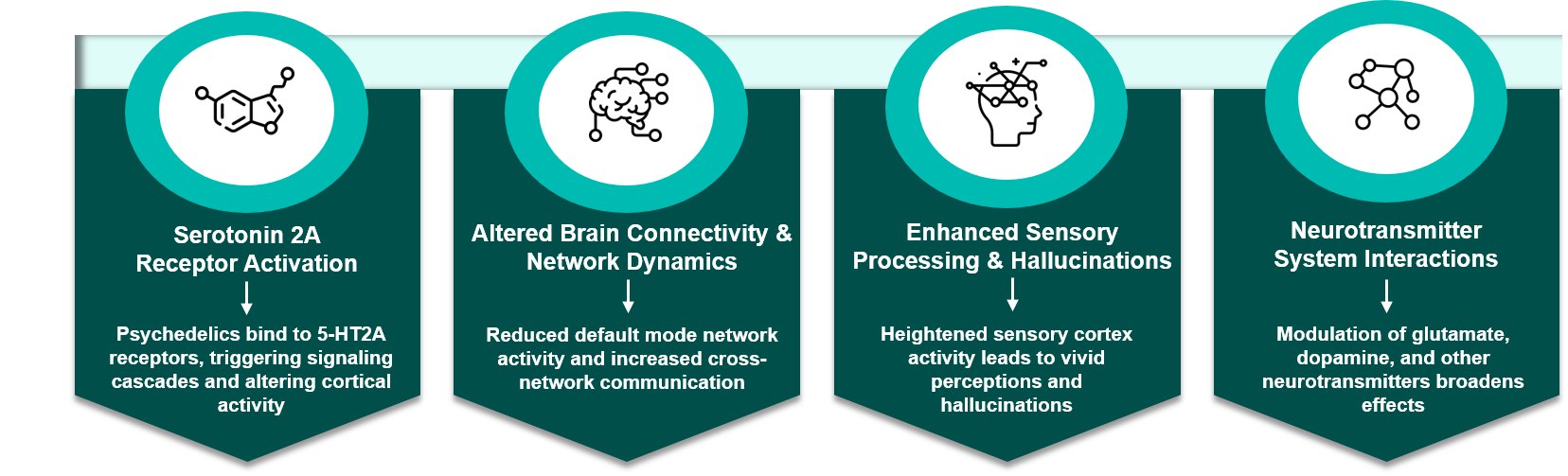
Most classic psychedelics—such as psilocybin, LSD, and DMT – primarily act by stimulating the serotonin 2A (5-HT2A) receptor, driving their hallucinogenic and consciousness-altering effects. Activation of these receptors re-alters brain connectivity, particularly disrupting the default mode network (DMN), which enables more fluid experiences of thought and identity. These also intensify sensory processing, hallucinations, synesthesia (mixing of the senses), and altered perception by altering the process of receiving sensory input. While serotonin signaling is central to classic psychedelics, other substances, such as MDMA and ketamine, exert their effects through other neurotransmitter systems (dopamine, glutamate), producing idiosyncratic subjective and therapeutic outcomes.
As the limitations of traditional treatments for mental health become increasingly evident, emerging next-generation psychedelics have gained urgency. The new wave of psychedelics is designed to deliver therapeutic benefits with distinct mechanisms, structures, and safety profiles.
- RE104: A prodrug of 4-OH-DiPT and with a structure very similar to psilocin, this psychedelic compound is derived from “magic mushrooms”. This new treatment for postpartum depression (PPD) has been developed by Reunion Neuroscience and offers a psychedelic experience over a shorter time frame.
- BPL-003: A synthetic form of Mebofotenin (a psychedelic occurring in many plants) has been developed by Beckley Psytech, wherein, following a single dose of BPL-003, 55% of participants had a rapid antidepressant effect the day following dosing.
- EMP-01: Expediting the progress of viable mental health treatment, Atai Life Sciences has developed a new product with features similar to MDMA (ecstasy) but offers less intense psychedelic effects, while providing a lower risk profile than MDMA and comparable therapeutic effects.
Clinical Pipeline: Momentum Building towards Approval Milestone
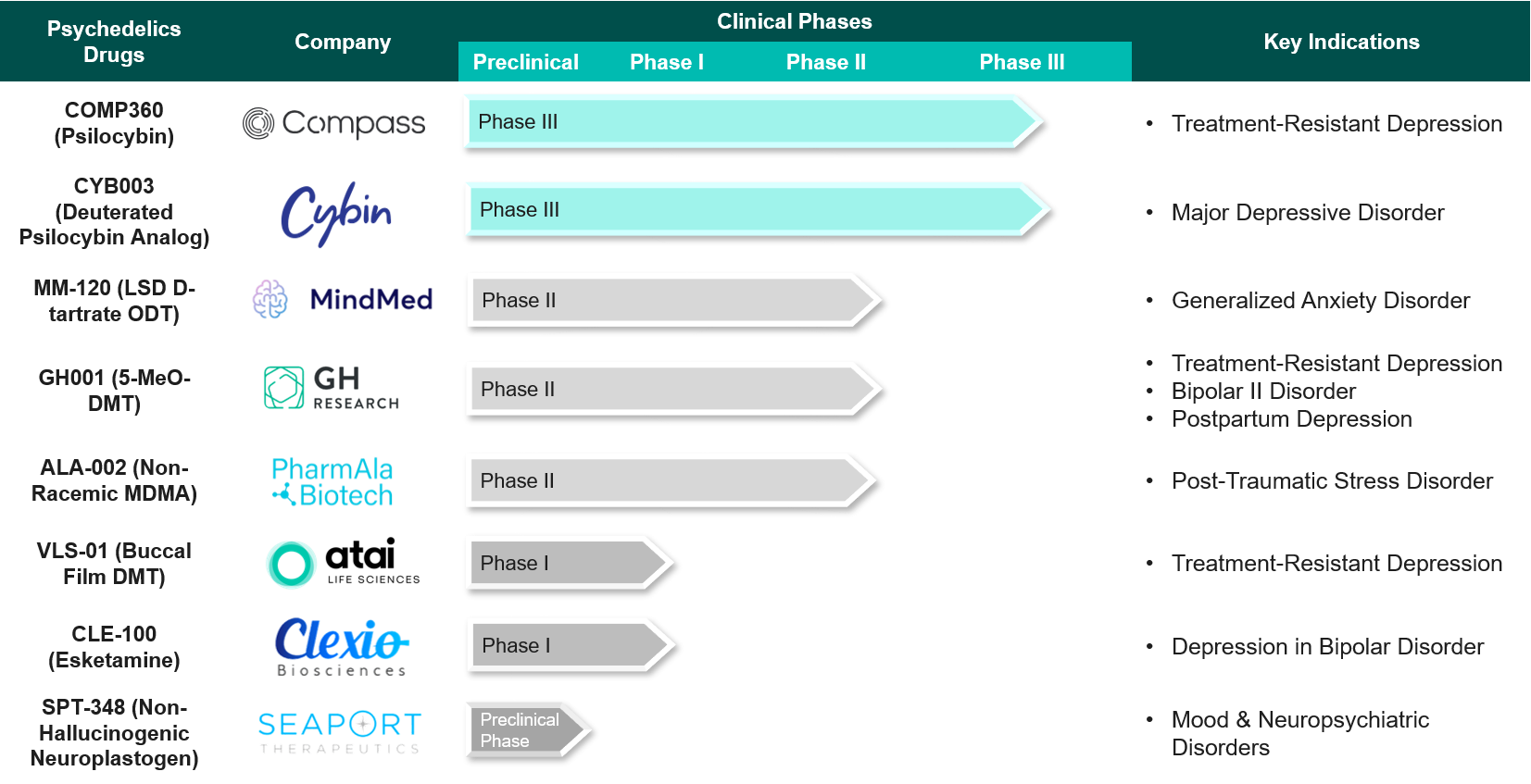
With several extensive and carefully planned trials aimed at disorders like depression, PTSD, addiction, and anxiety, the therapeutic development of psychedelics for mental health is rapidly accelerating. The clinical-stage pipeline is dominated by treatments for depression, with 23 candidates being developed for different types of depressive disorders. Alcohol and other drug use disorders are the second most researched condition. Anxiety and PTSD each have six potential investigators. There are 20 psilocybin trials and 14 DMT trials for different mental health conditions. Compass Pathways’ late-stage trial is one of the most closely watched, as success there could mark a turning point for regulatory approval. Other firms like Cybin, Awakn Life Sciences, and MindMed are pushing forward with diverse approaches, from proprietary formulations to combined psychotherapy protocols.
The convergence of rigorous trial design, growing public acceptance, and regulatory openness has made the current times more impactful. Positive late-stage results could allow us to see the
The convergence of rigorous trial design, growing public acceptance, and regulatory openness has made the current times more impactful. Positive late-stage results could allow us to see the first psychedelic-assisted therapies approved within the next few years—potentially reshaping mental health treatment paradigms.
Leadership Network & Strategic Priorities
The psychedelic mental health is driven by a mix of established biotech players, emerging startups, and leading universities, wherein a wave of start-ups and academic institutions is expanding the innovation frontier.

Strategic Activities:

In June 2025, Clearmind Medicine entered into an agreement with a leading consulting firm specializing in government and political affairs to help expand the regulatory and policy framework for psychedelic-based therapeutics in the United States.

In May 2025, Cybin partnered with Thermo Fisher Scientific to manufacture its CYB003 therapy in the U.S., for the purpose of Phase 3 trials and possible commercial production for the treatment of Major Depressive Disorder.

In April 2025, Psyence BioMed made a significant investment of $500,000 in PsyLabs, thus furthering the partnership and granting Psyence expanded exclusive rights to pursue pharmaceutical-grade psilocybin and first-refusal rights to Ibogaine for the development of substance use disorder treatments.

In March 2025, Monash University researchers published a study examining the clinical trial effects of MDMA-assisted psychotherapy for PTSD to help inform Australia’s first clinical guidelines for the use of MDMA in clinical practice.
Digital Synergies in Psychedelic Medicine
Platforms like iSTRYM collect real-time data on environmental conditions, heart rate, activity level, and mood, and further use artificial intelligence (AI) to actively provide new insights to physicians. AI-assisted drug discovery could lead to new psychedelic molecules that have therapeutic intent and none or reduced toxicity. For prospective patients considering psychedelic therapy, virtual therapists utilizing artificial intelligence-powered chatbots could offer the opportunity for initial screening, education, and support.
Regulatory Progression in Psychedelic Therapies
The last ten years have seen quick changes within the regulatory landscape around the use of psychedelics for mental health treatment, with some key fingerprints along the way. In June 2023, the F.D.A. published its first draft guidance for the design of clinical trials on psychedelic drugs. This was a major step in delivering to researchers the baseline advice around safety, efficacy, and complexity of paradigms for the use of these types of therapies. However, an F.D.A. Psychopharmacologic Drugs Advisory Committee voted against making a recommendation for MDMA-assisted therapy for PTSD in June 2024 because of issues of study design, potential bias, cardiovascular and abuse considerations, resulting in a formal rejection from the F.D.A. for MDMA-assisted therapy in August 2024 with a note that more clinical trials would need to be undertaken for safety and efficacy review. Despite the struggles for MDMA, other psychedelics such as psilocybin have begun to transition to late-stage clinical development, and there remains a robust amount of clinical research and regulatory paradigm around psychedelics.
Future Outlook and Conclusion
The future of psychedelics looks bright as research continues, regulatory changes are developed, and public perception has been quickly advancing. More and more, clinical trials are revealing that the compounds, such as psilocybin, MDMA, and novel-generation psychedelics, will provide rapid, sustained symptom relief from depression, PTSD, and addiction – especially when treatments have previously failed. As locations legalize and clinics specializing in psychedelic treatment emerge, access to safe and professionally guided psychedelic therapy is being expanded. With the continuous development of better drugs, delivery platforms, and personalized treatment, the opportunity to transform mental health treatment with psychedelics is paramount to create better outcomes for millions of individuals and society’s understanding of mental health and psychological well-being.
Stellarix Capabilities in Psychedelic Neurotherapeutics
The next decade will determine if psychedelic neurotherapeutics remain niche or specialized interventions or evolve into a foundational pillar of modern psychiatry. Active engagement today – through rigorous science, responsible innovation, and cross-sector collaboration will transform into tangible, real-world mental health impact tomorrow.
Stellarix assists stakeholders navigating through the emerging psychedelic neurotherapeutics landscape by integrating deep scientific expertise with strategic, regulatory, and commercialization expertise.
Our capabilities are designed to help organizations progress from initial exploration to confident, informed, and execution-ready decision-making.
Key Capabilities:
- Landscape & Horizon Scanning: Comprehensive mapping of psychedelic compounds, indications, mechanisms of action, and development pipelines across academia, startups, and industry.
- Clinical & Evidence Assessment: Evaluation of clinical trial maturity, durability of outcomes, safety profiles, and differentiation versus existing psychiatric therapies.
- Regulatory & Policy Insight: Analysis of evolving regulatory pathways, controlled substance frameworks, and global approval precedents shaping market entry.
- Market & Access Strategy: Assessment of care delivery models, reimbursement feasibility, clinician adoption barriers, and scalability constraints.
- Innovation & Partnership Strategy: Identification of high-value collaboration, licensing, and acquisition opportunities across the psychedelic ecosystem.
- Future-Readiness Planning: Strategic road-mapping to support long-term positioning as psychedelic therapies transition toward mainstream psychiatry.
Through an evidence-led and system-level approach, the Stellarix team enables informed, responsible participation in one of the most transformative shifts underway in mental health care.
Let's Take the Conversation Forward
Reach out to Stellarix experts for tailored solutions to streamline your operations and achieve
measurable business excellence.