Nrf2, Nuclear Factor Erythroid 2-related Factor, Activators
Currently, the market is shifting toward developing skincare products focused on oxidative stress, yet it is also saturated with antioxidant claims.
Here, Nuclear Factor Erythroid 2-related Factor 2 (Nrf2) activation serves as an upstream, fundamental mechanism for cytoprotection. Nrf2, encoded by the gene NFE2L2, serves as a central transcriptional regulator of cellular defense mechanisms against oxidative stress.
Nrf2 exerts its protective function by binding to antioxidant response elements (AREs) in the promoters of target genes, thereby inducing the transcription of a broad array of cytoprotective enzymes and proteins involved in detoxification, redox homeostasis, and oxidative stress mitigation.
Nrf2 shifts from merely managing symptoms to actively altering fundamental cellular defense mechanisms.
However, several challenges exist in harnessing Nrf2’s market potential: high cost, steep learning curve, uncertainties of the market landscape, and R&D complexities.
The main question is how companies can turn Nrf2 activation into a profitable product, given high R&D costs and other significant barriers.
In this regard, the article explores Nrf2 activators, their mechanisms of action, key innovations, and the technical barriers that hinder commercialization among CPG players.
How Nrf2 Works?
It is important to distinguish Nrf2 from Nuclear Respiratory Factor 2 (NRF-2), also referred to as GA-binding protein alpha (GABPA), which mainly plays a role in regulating mitochondrial biogenesis and cellular energy metabolism through its interaction with peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC1α).
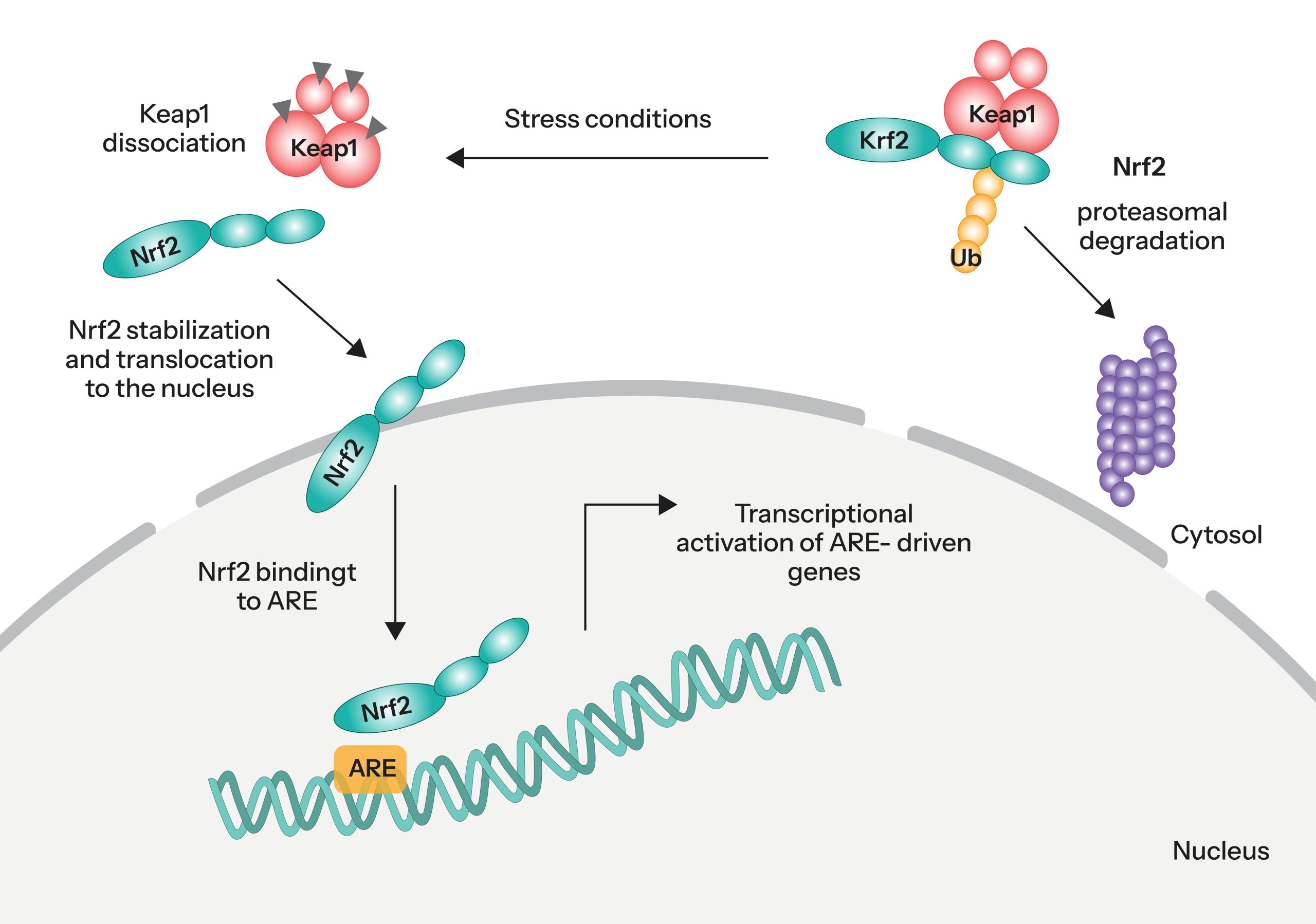
Under normal conditions, Nrf2 remains in an inactive state through its cytoplasmic association with Kelch-like ECH-associated protein 1 (Keap1), which facilitates its degradation. Upon exposure to oxidative stress, this interaction is disrupted, allowing Nrf2 to escape degradation, accumulate in the cytoplasm, and translocate into the nucleus.
Once in the nucleus, Nrf2 binds to antioxidant response elements (ARE) within the promoter regions of target genes, thereby initiating the transcription of a broad array of cytoprotective and detoxification genes. The molecular mechanism underlying this regulatory cascade is illustrated in Figure 1.
A diverse array of compounds activates the Nrf2 signaling pathway through distinct mechanisms. Electrophilic agents such as curcumin and sulforaphane activate Nrf2 by covalently modifying cysteine residues on KEAP1, thereby disrupting its ability to target NRF2 for proteasomal degradation.
Additionally, peptide mimetics like LDEETGEFL-NH₂ and small molecules such as benzenesulfonyl-pyrimidone inhibit the Nrf2–KEAP1 protein–protein interaction, stabilizing Nrf2 and promoting its translocation into the nucleus.
Notably, certain compounds, including Tideglusib, have been shown to activate Nrf2 independently of KEAP1, suggesting alternative regulatory pathways.
Despite these variations in acting mechanism, the majority of known NRF2 activators exert their effects via modulation of KEAP1, underscoring their central role in Nrf2 pathway regulation.
The functionality is just the beginning. Explore the complete guide, including the ingredient innovation pipeline, technical barriers to its widespread adoption, and its commercial landscape. Download the full article at:
Let's Take the Conversation Forward
Reach out to Stellarix experts for tailored solutions to streamline your operations and achieve
measurable business excellence.